When people think about environmental monitoring, they often focus on laboratory analysis and the final exposure data reported to customers. In reality, the reliability of that data depends on quality control measures that begin long before a sample is analyzed. At Assay Technology, quality control starts at the earliest stages of manufacturing and continues through every step that influences the final sampling product.
Environmental exposure measurements influence workplace safety decisions, regulatory compliance, and occupational exposure assessments. Because of this, the devices used to collect those measurements must be produced under controlled conditions that protect the integrity of the data they ultimately generate.
Environmental Monitoring as a Preventive Diagnostic
Assay products function as environmental diagnostics intended to prevent illness caused by chemical exposures. Similar to medical diagnostic tests such as blood tests, these measurements are frequently performed when workers are healthy in order to confirm that workplace conditions remain safe.
Because of this preventive role, the quality assurance and quality control systems used in manufacturing environmental monitoring products share similarities with those used in pharmaceutical and medical diagnostic manufacturing.
These systems focus on controlling both the materials used to create the product and the environmental conditions that could influence product performance.
Quality Assurance and Quality Control
Quality Assurance (QA) and Quality Control (QC) address different aspects of the manufacturing process.
Quality Assurance generally refers to the policies, procedures, and systems that define how a product should be manufactured. Quality Control refers to the specific performance tests conducted during and after each stage of production.
Together, these systems ensure that products are manufactured consistently and that any deviation capable of influencing performance can be detected and corrected before the product reaches customers.
Good Manufacturing Practices
In the medical manufacturing field, quality systems are commonly structured around Good Manufacturing Practices (GMP). These practices require that every ingredient and every process step capable of influencing product performance be evaluated and controlled before it enters the manufacturing process.
Although formal GMP regulations do not specifically apply to workplace health and safety sampling devices, Assay Technology follows many of the same principles. This approach reflects both the company’s internal standards and the experience of employees who previously worked in medical manufacturing environments where these practices are standard.
Applying these principles reduces variability in the manufacturing process and improves the reliability of the monitoring devices used to generate exposure data.
Testing and Control at Multiple Stages
One principle of GMP systems is that testing should occur at multiple stages of production rather than only at the final stage.
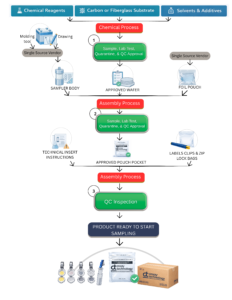
For Assay products, testing and control occur across three primary stages:
- Raw materials
- Sampling Media
- Pouched (final) products
Each stage includes verification tests before the process is allowed to continue.
This staged approach ensures that potential issues are detected early and prevents defective materials or intermediate products from progressing further in the manufacturing process.
Testing Doesn’t End at Production
Even after products leave the manufacturing line, quality control continues. Assay Technology performs additional verification testing on many products during their shelf life, including mid-shelf and end-of-shelf quality checks.
This extended monitoring helps confirm that the products continue to perform as expected over time and that the exposure measurements they support remain reliable for the entire duration of their intended use.
This approach reflects a simple principle: reliable exposure data requires ongoing verification, not just testing during manufacturing.
Following a Defined Manufacturing Process
Consistency in manufacturing requires that every step be performed according to a documented procedure.
At Assay Technology, this process is defined in a document known as the Master Formula (MF). The Master Formula specifies the ingredients used to manufacture the product as well as the sequence of process steps required to produce it.
Following a defined manufacturing process ensures that products are produced consistently across batches and that deviations can be identified and investigated.
Controlling Environmental Contamination
Product quality can be influenced not only by the materials and steps used during manufacturing but also by environmental conditions surrounding the production process.
The Master Formula therefore includes procedures intended to prevent extrinsic environmental contamination.
Extrinsic contamination refers to environmental factors that could affect product integrity if not controlled during manufacturing.
Assay’s manufacturing specifications include both preventive steps designed to minimize environmental contamination and tests intended to detect whether contamination may have occurred during any stage of production.
These controls help ensure that environmental monitoring devices measure workplace exposure conditions rather than contamination introduced during manufacturing.
Why Data Integrity Matters
Industrial hygienists and environmental health and safety professionals rely on monitoring data to evaluate airborne hazards, determine worker exposure levels, and assess whether workplace controls are effective. The reliability of those decisions depends on the accuracy and traceability of the data used to support them.
For this reason, the integrity of environmental monitoring products must be maintained throughout the entire manufacturing process, beginning with raw materials and continuing through final product packaging.
Sample Till You’re Sure
Environmental monitoring is not simply about collecting samples or generating laboratory results. It is about producing reliable exposure data that can be used to protect worker health and support defensible safety decisions.
Every control, inspection, and verification step performed during manufacturing exists to support that goal.
At Assay Technology, the philosophy behind these systems is simple:
Sample till you’re sure.